
Metabo
Description
21 partners are involved in the METABO project, from 9 different UE countries, including hospitals, universities and large companies, principally from Italy, Greece and Spain. MEDTRONIC, world leader in medical technology, is the general coordinator of the consortium. The aim of the METABO project is the improvement of clinical management of diabetes, using the latest available technology to define new methods of patient follow-up within the health service, and to help those with diabetes to improve their own management of the illness.
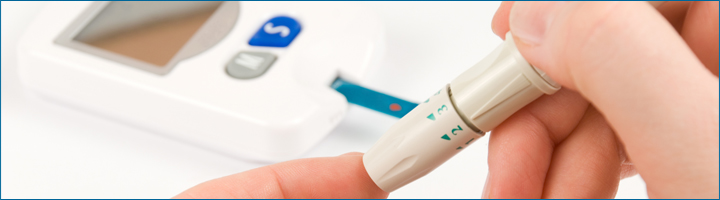
The METABO platform will concentrate on tracking people suffering from diabetes, mellitus, types 1 and 2. The solution that METABO proposes will affect both medical professionals dedicated to primary and specialised care, and the patients themselves, offering tools facilitating communication between the various people involved. This will be achieved through the development of a technological platform which connects the different factors implicated in collecting the relevant information and the process of data and diagnosis support, thereby permitting the patient and the medical team to manage the illness more efficiently, giving access to a higher level of information in comparison with current medical practice.
The types of parametres that will be the object of said follow-up, besides the traditional clinical parametres, will include the concentration of subcutaneous glucose, energy used through physical activity, blood pressure and weight. During the follow-up the patient’s lifestyle, dietary habits, medication and its effects, physical activity and the subsequent changes to daily life will also be monitored.
The ITACA-TSB group is responsible for developping communication using medical monitoring sensors, and also for assisting in the development of applications for monitoring lifestyles.


